
A recent study has revealed fascinating findings about the multi-kingdom composition of bacterial biofilms. A mixture of bacterial and fungal species, these "superorganisms" possess greater resistance to antibacterial agents and higher virulence due to their unique leaping ability.
By Dr. Mehmood Asghar PhD., M Phil, BDS
Despite meaningful improvements in oral healthcare facilities worldwide, dental caries continues to be a global concern causing significant healthcare and financial impact (Wen et al., 2022). For example, it has been shown that over 500 million children worldwide have untreated caries in their primary teeth (Uribe et al., 2021). Similarly, according to the 2015 Global Burden of Disease Study, dental caries ranks the first for permanent teeth, affecting 2.3 billion people globally (Wang et al., 2016). Hence, research continues to identify the pathophysiology of dental caries, so that novel, cost-effective, and minimally invasive treatment options can be developed to curb the global problem.
Dental caries is the localized destruction of the dental hard tissues caused by the release of acidic by-products during bacterial fermentation of carbohydrates in the oral cavity (Wen et al., 2022). The formation of biofilms on the surfaces of teeth is regarded as the most crucial requirement in the initiation and progression of dental caries, which refers to the adherence of multicellular structures on biological surfaces such as the teeth (Lohse et al., 2018). The current body of knowledge indicates that bacterial- and fungal-induced biofilms can significantly impact oral health.
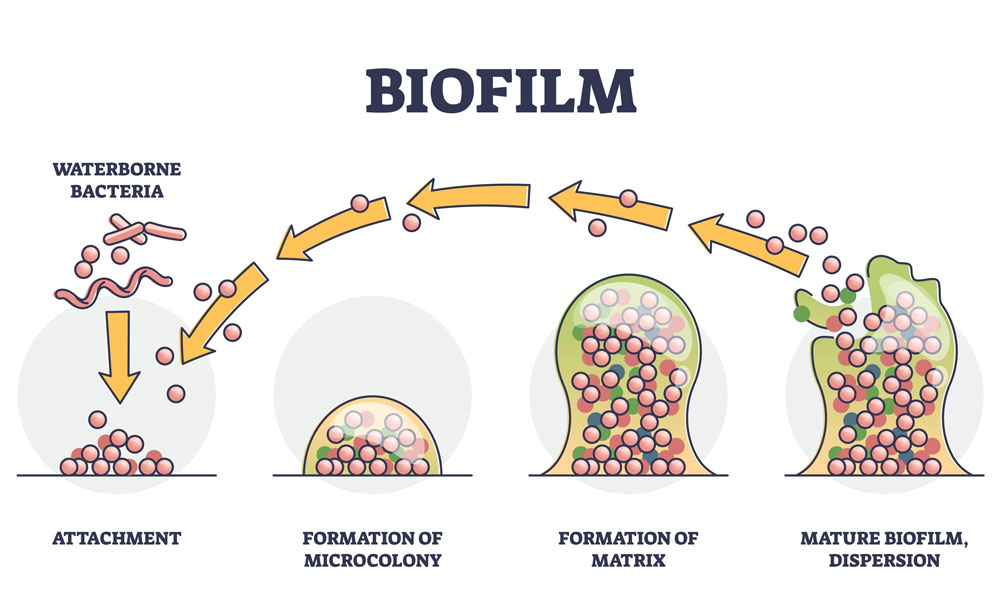
Previously, it was known that the higher incidence of Streptococcus mutans and Candida albicans in the saliva resulted in increased dental caries. Furthermore, previous studies have shown that the interaction of these two species resulted in the formation of interkingdom biofilms with enhanced virulence under sugar-rich oral conditions (Falsetta et al., 2014; Hwang et al., 2017). However, the underlying mechanism of forming these interkingdom biofilms and the reason behind their enhanced virulence were not fully known.
A recent study (Ren et al., 2022) published in the Proceedings of the National Academy of Science, comprising researchers from the University of Pennsylvania School of Dental Medicine, has provided highly interesting findings about the cross-kingdom partnership between oral bacteria and fungi to form a “superorganism” with enhanced resilience and strength.
Using real-time multiscale imaging and computational analysis, the researchers discovered that bacterial clusters could attach to the yeasts and hyphal complexes, resulting in assemblages with superior surface colonization and growth rate, adhesion, and antibacterial and biofilm degradation resistance.
The Superorganisms: What is the Underlying Mechanism?
Using various imaging techniques, the researchers discovered that the saliva from children with dental caries enriched bacterial-fungal assemblages. In contrast, salivary samples from caries-free children only had single-cell bacteria or bacterial aggregates. Interestingly, it was observed that the salivary samples from caries-affected children were primarily comprised of S. mutans and C. albicans, corroborating the findings of previous studies.
Furthermore, α-Glucans, an extracellular polysaccharide mainly produced by S. mutans, was also detected in caries-affected salivary samples. The authors have proposed a possible role of α-Glucans in forming these bacterial-fungal assemblages. This was confirmed by the impairment of forming these bacterial-fungal assemblages when the supply of carbohydrates was discontinued under laboratory conditions.
Bacterial-fungal Assemblages in Saliva: Physical Features
Using fluorescence and 3D imaging, the researcher observed that these assemblages had fungal hyphae located at the periphery with surface adhesion in a pillar-like fashion. On the other hand, the bacterial cell clusters were mainly observed in the center, attached to the fungal hyphae like a “cargo.”
Using computational analysis, it was further revealed that the core of this superorganism comprised mainly S. mutans and C. albicans, while the periphery mostly had fungal hyphae extending outward. These coassembled species colonized the hydroxyapatite (HA) surfaces as a highly structured group. Interestingly, the shear force required to detach these coassembled biofilms was significantly higher than that needed for either species alone, with single-cell S. mutans strains showing the least resistance. This again shows a symbiotic relationship between this bacterial-fungal partnership.
Bacterial-fungal Assemblages in Saliva: Antibacterial Resistance
Further evidence of the symbiotic relationship between these assemblages was obtained when it was shown that they were significantly more resistant to chlorhexidine than either of the mono-species biofilms, with only minor neutralization of the peripheral cells of the multi-kingdom structure after a five-minute exposure. It was also revealed that the higher antibacterial resistance of these assemblages was due to the protective role of α-glucan, which provided a glue-like effect.
When a bacterial species other than S. mutans was used to induce co-assemblage, it was shown that such colonization had weak mechanical properties and antibacterial resistance. Hence, S. mutans and C. albicans are essential for forming these robust superorganisms. The peripheral fungal coating was also proposed to provide additional protection to the underlying bacterial core.
Bacterial-fungal Assemblages with Walking- and Leaping-like Functionalities
The researchers of this study observed a unique migratory behavior of these interkingdom biofilms. It was shown that a “leading edge” was formed in the assemblage that moved with time. This movement was brought about primarily by the fungal hyphae, while the bacterial core “hitchhiked” along with it. This finding was corroborated by destroying the fungal hyphae using an anti-fungal agent and observing that the movement functionality of the co-assemblage was significantly impaired.
Clinical Significance of These Findings
The findings of this study highlight the following:
- The existing body of knowledge suggesting the primary role of S. mutans and C. albicans in caries initiation and progression has also been corroborated by this study.
- The bacterial-fungal co-assemblages are more virulent, mechanically more robust, and antibacterial-resistant than single-cell biofilms.
- These multi-kingdom biofilms possess translational capabilities that allow them to cover a larger surface area, causing more disease progression in less time.
Future Implications
Further studies should focus on developing novel mechanisms to prevent or disrupt such multi-kingdom assemblages on dental tissues. Furthermore, there is a need to establish antibacterial- and anti-biofilm agents with enhanced virulence against such assemblages. This will pave the way forward in reducing dental caries' global financial and healthcare burden.
Author: Dr. Mehmood Asghar is a dentist by profession and an Assistant Professor of Dental Biomaterials at the National University of Medical Sciences, Pakistan. Dr. Asghar received his undergraduate and postgraduate dental qualifications from the National University of Science and Technology (NUST). He is also currently pursuing a Ph.D. in Restorative Dentistry from Malaysia. Apart from his hectic clinical and research activities, Dr. Asghar likes to write evidence-based, informative articles for dental professionals and patients. Dr. Asghar has published several articles in international, peer-reviewed journals.
References
Falsetta, M. L., Klein, M. I., Colonne, P. M., Scott-Anne, K., Gregoire, S., Pai, C.-H., Gonzalez-Begne, M., Watson, G., Krysan, D. J., & Bowen, W. H. (2014). Symbiotic relationship between Streptococcus mutans and Candida albicans synergizes virulence of plaque biofilms in vivo. Infection and immunity, 82(5), 1968-1981.
Hwang, G., Liu, Y., Kim, D., Li, Y., Krysan, D. J., & Koo, H. (2017). Candida albicans mannans mediate Streptococcus mutans exoenzyme GtfB binding to modulate cross-kingdom biofilm development in vivo. PLoS pathogens, 13(6), e1006407.
Lohse, M. B., Gulati, M., Johnson, A. D., & Nobile, C. J. (2018). Development and regulation of single-and multi-species Candida albicans biofilms. Nature Reviews Microbiology, 16(1), 19-31.
Ren, Z., Jeckel, H., Simon-Soro, A., Xiang, Z., Liu, Y., Cavalcanti, I. M., Xiao, J., Tin, N.-N., Hara, A., Drescher, K., & Koo, H. (2022). Interkingdom assemblages in human saliva display group-level surface mobility and disease-promoting emergent functions. Proceedings of the National Academy of Sciences, 119(41), e2209699119. https://doi.org/doi:10.1073/pnas.2209699119
Uribe, S. E., Innes, N., & Maldupa, I. (2021). The global prevalence of early childhood caries: A systematic review with meta-analysis using the WHO diagnostic criteria. International Journal of Paediatric Dentistry, 31(6), 817-830. https://doi.org/https://doi.org/10.1111/ipd.12783
Wang, H., Naghavi, M., Allen, C., Barber, R. M., Bhutta, Z. A., Carter, A., Casey, D. C., Charlson, F. J., Chen, A. Z., Coates, M. M., Coggeshall, M., Dandona, L., Dicker, D. J., Erskine, H. E., Ferrari, A. J., Fitzmaurice, C., Foreman, K., Forouzanfar, M. H., Fraser, M. S., . . . Murray, C. J. L. (2016). Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: a systematic analysis for the Global Burden of Disease Study 2015. The Lancet, 388(10053), 1459-1544. https://doi.org/https://doi.org/10.1016/S0140-6736(16)31012-1
Wen, P. Y. F., Chen, M. X., Zhong, Y. J., Dong, Q. Q., & Wong, H. M. (2022). Global Burden and Inequality of Dental Caries, 1990 to 2019. Journal of Dental Research, 101(4), 392-399. https://doi.org/10.1177/00220345211056247




